- Home
- Details
- Registry
- RSVP
- Strider knives san marcos
- Lego airplane
- Immortals fenyx rising switch review
- Grim dawn gamepedia
- Space runaway ideon online
- Free online pinochle games
- Series periodic table chemistry
- Microsoft office 2008 mac cheap
- Papyrus font generator
- Find my iphone alert
- Itpv extreme pro
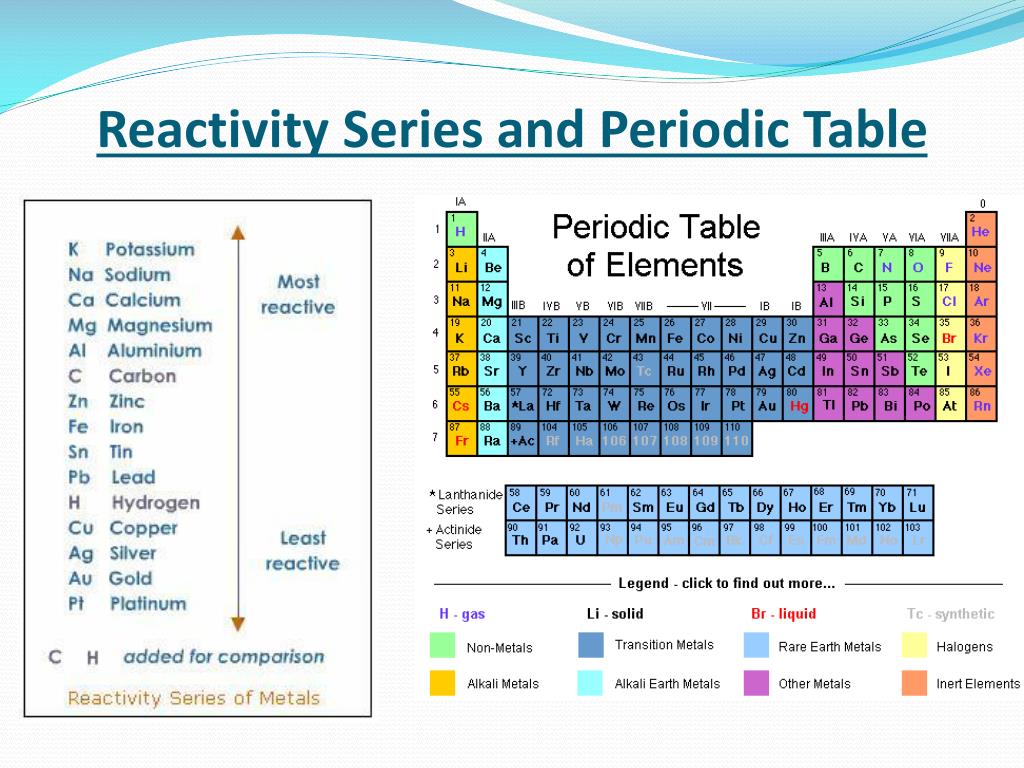
The aufbau (build up) principle and the electronic configuration of atoms provide a theoretical foundation for the periodic classification. Classification of elements on the basis of their electronic configuration: The seventh period is exact repetition of this scheme involving 5f orbitals. So, they are placed separately at the bottom of the table. Placing of these elements in the periodic table would require 14 extra groups this would make the table unnecessarily long.
SERIES PERIODIC TABLE CHEMISTRY SERIES
So, 4f is the first series of 14 inner transition elements. The sixth period is further complicated by the filling of 4f orbitals after 4d.f orbital can accommodate 14 electrons. The fifth period similarly contains 18 elements in 5s, 4d, 5p orbitals. Once these d-orbitals are filled, the three 4p orbitals are filled, and now 6 more elements are in this period. These 10 elements form the first transition series, occupying the ten middle groups of the periodic table. 10 electrons may be placed in these orbitals, each leading to a new element. The 3d orbitals appear next in the increasing order of energy. So, a new period, the forth one, starts with K, 4S1. This is why K is the next element after Ar, bears its outer most electron in 4s. The 4s orbital has lower energy than 3d orbitals. The scheme is exactly repeated inn third period. There are thus total (2+6) or 8 electrons in the second period. Addition of each electron corresponds to a new element. There are one 2s and three 2p orbitals, each of which can accommodate two electrons. The second period starts with the element having one electron in 2s shell, Li (2s1). The 1s orbital can accommodate only two electrons the first period contains only two elements, hydrogen (1s 1) and helium (1s 2). For hydrogen this is 1s lithium 2s, sodium 3s and so on. Each period starts with an element having one valance electron in the s-orbital. The arrangement of long periodic table may be very easily understood from the distribution of electrons in the various quantum levels. Elements having similar outer electronic configurations in their atoms are arranged in vertical columns, referred to as groups or families. The horizontal rows (which Mendeleev called series) are called periods and the vertical columns, groups. The arrangement of the table is consistent with the electron distribution in a Bohr-Sommerfeld atom model This is why the table is also referred to as the Bohr table.Ī modern version, the so-called “long form” of the Periodic Table of the elements, is the most convenient and widely used. A more modern arrangement was presented by Bury in 1921. The long form of the periodic table was introduced in a simple form by Rang in 1893 and by Werner in 1905. The physical and chemical properties of the elements are periodic functions of their atomic numbers. He understood that some of the elements did not fit in with his scheme of classification if the order of atomic weight was strictly followed. In particular, Mendeleev relied on the similarities in the empirical formulas and properties of the compounds formed by the elements.

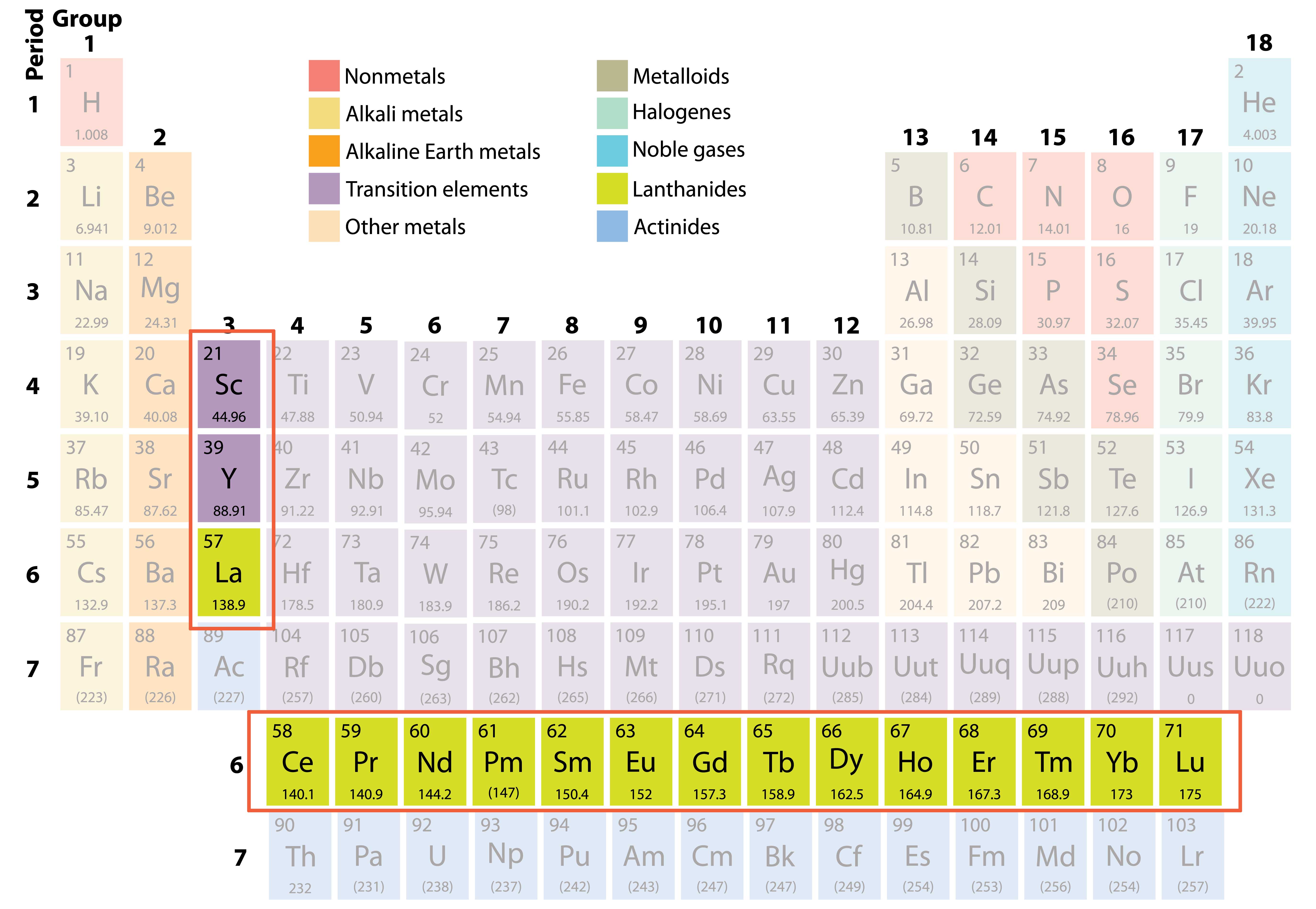
He fully recognized the significance of periodicity and used broader range of physical and chemical properties to classify the elements. The properties of the elements are a periodic function of their atomic weights.
- Home
- Details
- Registry
- RSVP
- Strider knives san marcos
- Lego airplane
- Immortals fenyx rising switch review
- Grim dawn gamepedia
- Space runaway ideon online
- Free online pinochle games
- Series periodic table chemistry
- Microsoft office 2008 mac cheap
- Papyrus font generator
- Find my iphone alert
- Itpv extreme pro
